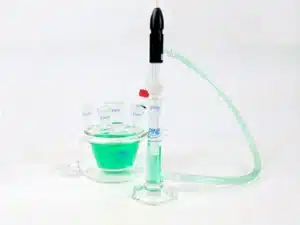
1. Description and Kit Contents
The Reference Electrode Salt Bridge Kit physically and diffusionally isolates a reference electrode from the sample electrolyte. This helps ensure proper operation of the reference electrode. The kit consists of a fritted tube probe, a 14/20 adapter for the probe, a 14/20 glass adapter, a flexible tube salt bridge, an external reservoir, a rubber stopper, and a 60 mL size pipette bulb. The basic principle of the kit is to isolate the reference electrode in the External Reservoir and complete the electrical connection to the cell through the electrolyte in the Flexible Tube Salt Bridge and the Fritted Tube Probe.
2. When to Use a Salt Bridge
An ideal reference electrode provides a stable potential and is not affected by environmental factors. The stability of a practical reference electrode requires that the sample electrolyte does not contain components that might interfere with the redox chemistry of the reference electrode. In many cases, interference is minimized through separation of the reference electrode and sample electrolyte by a porous frit. The frit, with its high porosity and small pores, serves to maintain ionic conduction between the reference electrode and sample electrolyte while impeding the convective transport of components through the frit. In other cases, however, the sample electrolyte or experimental conditions affect the reference electrode potential despite the presence of the porous frit. In these cases, a salt bridge must be added to isolate the reference electrode from the sample electrolyte.
To illustrate when a salt bridge is desirable, the silver chloride reference electrode will be considered because it is one of the most widely used reference electrodes for aqueous systems. Limitations for the silver chloride reference electrode include shifting liquid junction potential as temperature changes, frit clogging, and contamination of the sample with chloride ions. If these factors are a concern, a salt bridge should be used. These limitations are discussed more below:
- Temperature Change. The liquid junction potential is sensitive to change in temperature. Therefore, if the main cell is heated or cooled, researchers should try to minimize the effects of temperature on the reference electrode. The reference electrode can be placed outside the main cell and kept at the room temperature if a salt bridge is utilized.
- Frit Clogging. In some cases, the sample electrolyte contains chemicals which will react with the reference electrolyte to form insoluble compounds at the frit interface. For example, if the sample electrolyte contains a high concentration of perchlorate ion,
, then,
ion leaked through the frit can react with
, forming
, a salt with a low solubility in water. The
salt deposits in the pores of the frit on the reference electrode tip and causes liquid junction potential variations. To solve this problem, a salt bridge can be added and filled with
or
, both of which are compatible with
and
.
- Chloride or Water Sensitive Reactions. A porous frit avoids the convective transport of electrolyte components but over time, the components will inevitably leak out through diffusional transport. For many catalysis reactions, even a small leakage of reference electrolyte has detrimental effects on catalytic efficiency. Many reactions are sensitive to components (water or chloride ions from saturated KCl or NaCl) in reference electrode fill solutions. A salt bridge serves to separate the sensitive reaction of interest from the reference electrode compartment.
While only a silver chloride reference was discussed above, the limitations described can be applied to all reference electrodes with their solvents and ion reactivity modified to fit the electrochemical system under study.
3. Salt Bridge Assembly Instructions
Follow these steps to use the Reference Electrode Salt Bridge Kit.
3.1. Assemble the Kit Pieces
Slide the 14/20 PTFE Probe Adapter over the Fritted Tube Probe. Loosely tighten the red cap to hold the Probe Adapter in place. Connect one end of the Flexible Tube Salt Bridge to the barbed hose connector on the Fritted Tube Probe. Connect the other end of the Flexible Tube Salt Bridge to the barbed hose connector of the External Reservoir.
3.2. Attach the Kit to an Electrochemical Cell
Mate the Fritted Tube Probe onto the 14/20 port of a glass electrochemical cell. Adjust the height of the Fritted Tube Probe by untightening the red cap of the 14/20 PTFE Probe Adapter and sliding the Fritted Tube Probe into place; the tip of the Fritted Tube Probe should be at least 1 inch below the sample liquid. Tighten the red cap when the desired height is reached.
3.3. Fill the Salt Bridge
Recall that various situations give rise to the use of a salt bridge. If a salt bridge is needed due to temperature change or sensitivity to water or chloride ions, the sample electrolyte can be used to fill both the salt bridge and the electrochemical cell. If a salt bridge is needed due to frit clogging, an electrolyte compatible with both the reference electrode and the sample electrolyte should be used to fill the salt bridge. In both cases, the electrolyte inside the salt bridge must be conductive. However, the two situations require two different methods to fill the salt bridge.
3.4. Filling the Salt Bridge with the Sample Electrolyte
Attach the Pipette Bulb to a greased 14/20 Glass Adapter. Squeeze the Pipette Bulb to deflate it, and continue holding it in its deflated state. With your other hand, raise the External Reservoir to the highest point possible with respect to the electrochemical cell; the Flexible Tube Salt Bridge should be nearly vertical. Attach the deflated Pipette Bulb with 14/20 Glass Adapter to the 14/20 port of the External Reservoir and slowly remove your hand from the bulb. This will apply suction (negative pressure) at the 14/20 port of the External Reservoir and electrolyte liquid should enter the External Reservoir from the electrochemical cell.
When the reservoir is filled, lower the External Reservoir to the same height as the electrochemical cell; the liquid level inside the reservoir should reach the same level as that in the glass cell, about 1 to 2 inches. The Pipette Bulb should still be attached to the External Reservoir.
Remove the Pipette Bulb and 14/20 Glass Adapter from the External Reservoir. If desired, wipe away the grease by using a towel wetted with a solvent like pentane. Insert the external reference electrode into the reservoir.
3.5. Filling the Salt Bridge with an Electrolyte different from the Sample Electrolyte
The directions to fill the salt bridge with a different electrolyte are very similar to those previously outlined, except that an additional piece of glassware with a 14/20 port is needed. Pour the salt bridge electrolyte into the new piece of glassware, fill the salt bridge, and mount the reference electrode as described above. Fill the main electrochemical cell with sample electrolyte and make sure that there is an unoccupied 14/20 port available. Remove the Fritted Tube Probe from the salt bridge electrolyte, quickly rinse the outside with the sample electrolyte before mounting it on the 14/20 port of the main electrochemical cell.
4.5. Disassembly and Clean Up Instructions
Remove the reference electrode from the External Reservoir, rinse with copious solvent, and immerse the frit in a proper storage solution. Raise the External Reservoir so that its bottom is above the barbed hose connector of the Fritted Tube Probe. The electrolyte in the salt bridge will drain into the main electrochemical cell. Alternatively, to avoid contamination of the main cell with the liquid in the reservoir, you may drain the salt bridge by pointing the tip of the Fritted Tube upward and allowing all liquid to flow back to the External Reservoir.
Properly dispose of any unused liquid in the above procedure. Take apart the Reference Electrode Salt Bridge Kit and rinse the External Reservoir, Flexible Tube Salt Bridge, and Fritted Tube Probe with copious solvent. Rinse other kit pieces to desired cleanliness, and store all parts in the kit dry.