General Electrochemistry
Back to General Electrochemistry Back to Applications Back to Knowledgebase HomeElectrode Shroud Materials Overview
Last Updated: 5/7/19 by Neil Spinner
1Abstract
Chemically-stable shroud materials that operate within a desired temperature range are essential to perform any electrochemical study. Pine Research offers various electrode shroud materials including polytetrafluoroethylene (PTFE), polychlorotrifluoroethylene (PCTFE), and polyether ether ketone (PEEK). The article herein aims to introduce these electrode shroud materials and detail their compatibility with different systems.
2Material Source Disclaimer
The shroud of an electrode is designed to cover electrical components and prevent accidental contact with live components. Therefore, inert, non-conductive materials are used to construct an electrode shroud. Common shroud materials include polymers like polytetrafluoroethylene (PTFE), polychlorotrifluoroethylene (PCTFE), and polyether ether ketone (PEEK) (see Table 1).
Like many polymers, the physical properties of PTFE, PCTFE, and PEEK are directly affected by their method of fabrication. For example, the molding and sintering conditions used to produce PTFE affect its overall molecular weight, alignment of polymer chains, percent crystallinity, macroscopic flaws, and microporosity; these factors influence the permeability, stiffness, resiliency, and impact strength of PTFE.
 DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
Pine Research uses polymers from several vendors with varying fabrication methods to shroud its electrodes. For this reason, Pine Research offers specific operating conditions under which its materials perform (provided herein).
DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
Pine Research uses polymers from several vendors with varying fabrication methods to shroud its electrodes. For this reason, Pine Research offers specific operating conditions under which its materials perform (provided herein).
| Shroud Material | Chemical Formula | Chemical Structure |
| Polytetrafluoroethylene (PTFE) | 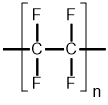 |
|
| Polychlorotrifluoroethylene (PCTFE) | 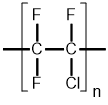 |
|
| Polyether Ether Ketone (PEEK) | 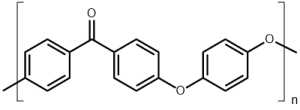 |
Table 1. Chemical Formula and Structure of Shroud Materials
3Polytetrafluoroethylene (PTFE)
3.1Physical Properties
PTFE is a fluoropolymer that is produced when tetrafluoroethylene (C2F4) undergoes free-radical polymerization (see Equation 1). The formation of PTFE’s strong carbon-carbon bonds and very strong carbon-fluorine bonds render it unreactive; only alkali metals and gaseous fluorine are known to break carbon-fluorine bonds under most conditions, but fluorinating agents like xenon difluoride or other metals like aluminum and magnesium can also break the bonds at higher temperatures. Additionally, the fluorine atoms form a protective sheath around the carbon atoms, giving rise to low permeability of water and other liquids.
| (1) |
Once in its polymerized state, PTFE is white and waxy. Due to its high microporosity, the number and size of voids created by imperfect particle fusion, PTFE has a high dielectric constant, making it amenable to use as an insulator between electrical components.
 DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
Its low thermal expansion, low coefficient of friction, high hydrophobicity, and high melting point also render it useful as a non-stick coating for cookware.
DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
Its low thermal expansion, low coefficient of friction, high hydrophobicity, and high melting point also render it useful as a non-stick coating for cookware.
3.2Practical Use as a Shroud Material
When selecting an electrode shroud material, researchers must consider the chemical compatibility, operating temperature range, and mechanical properties of the shroud and their system of study.
3.2.1Chemical Compatibility
A researcher may choose to work with an electrode that has a PTFE shroud if they are working with reactive and corrosive materials. PTFE shroud material can be used for both aqueous and non-aqueous work, and is the most chemically-resistant shroud material that Pine Research offers.
3.2.2Thermal Stability
PTFE melts at high temperature (327°C ) and has relatively low linear thermal expansion coefficients at temperatures below 25°C (see DuPont’s Properties Handbook
 DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
for more information). An electrode with a PTFE shroud cannot be thermally cycled to temperatures as high as its melting point, however, because the thermal expansion coefficient of PTFE increases nonlinearly as temperature increases. As a result, larger, nonlinear volume changes occur in PTFE shroud materials at higher temperature. Significant volume changes can cause protrusion of the shroud from the electrode edge and/or a leaky seal between the electrode and shroud material. As part of the quality assurance protocol, Pine Research tests all fixed disk/ring electrodes at ambient (room) temperature and has approved PTFE-shrouded electrode use from 10°C to 25°C. Exposing an electrode with a PTFE shroud to temperatures outside this range may create a leak between the electrode material and the shroud and is not recommended by Pine Research.
DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
for more information). An electrode with a PTFE shroud cannot be thermally cycled to temperatures as high as its melting point, however, because the thermal expansion coefficient of PTFE increases nonlinearly as temperature increases. As a result, larger, nonlinear volume changes occur in PTFE shroud materials at higher temperature. Significant volume changes can cause protrusion of the shroud from the electrode edge and/or a leaky seal between the electrode and shroud material. As part of the quality assurance protocol, Pine Research tests all fixed disk/ring electrodes at ambient (room) temperature and has approved PTFE-shrouded electrode use from 10°C to 25°C. Exposing an electrode with a PTFE shroud to temperatures outside this range may create a leak between the electrode material and the shroud and is not recommended by Pine Research.
3.2.3Mechanical Properties
Hardness and modulus are two common mechanical properties that researchers should consider before selecting a shroud material. The hardness of a material reflects its resistance to permanent indentation and is often measured using the Shore method. In the Shore method, the penetration depth of a pressurized, spring-loaded needle into a material is recorded and set proportional to the hardness; a scale between 0 and 100 is used, with higher values indicating a harder material. PTFE is a fairly soft material, with a Shore D (durometer) hardness of ~55.
 DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
Under the rigors of electrochemical research; e.g., routine polishing with aluminum oxide microparticle slurries as suggested by Pine Research,
DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
Under the rigors of electrochemical research; e.g., routine polishing with aluminum oxide microparticle slurries as suggested by Pine Research,
 Pine Research Electrode Polishing Guide
the PTFE shroud will easily polish with the electrode material. In one way, this is advantageous since the disk/ring and shroud will stay coplanar after many polishing preparations. However, the low hardness of PTFE also dictates that care must be used not to damage, scrape, or dent the PTFE shroud material.
Pine Research Electrode Polishing Guide
the PTFE shroud will easily polish with the electrode material. In one way, this is advantageous since the disk/ring and shroud will stay coplanar after many polishing preparations. However, the low hardness of PTFE also dictates that care must be used not to damage, scrape, or dent the PTFE shroud material.
For most materials, a linear correlation between hardness and mechanical strength exists. Modulus is a common mechanical strength property, and is defined as a material’s resistance to deformation. It is measured by dividing the initial stress exerted upon the material by ΔL/L, where ΔL/L is the fractional increase in the material’s length. There are several types of moduli values for a material including tensile, compressive, and flexural. For many researchers, the modulus of an electrode shroud bears no significant role. However, for researchers who work in high pressure or mechanical stress environments, only materials with high moduli should be used. PTFE has a low tensile modulus of 0.5 GPa and should not be used in high mechanical stress applications as it will deform easily (for more information on PTFE modulus values, see Dupont's Properties Handbook).
 DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
4Polychlorotrifluoroethylene (PCTFE)
4.1Physical Properties
PCTFE is a chlorofluoropolymer that is produced when chlorotrifluoroethylene (C2ClF3) undergoes free-radical polymerization (see Equation 2). The formation of PCTFE’s strong carbon-carbon bonds and very strong carbon-fluorine bonds render it mostly unreactive like PTFE. Replacement of a carbon-fluorine bond with a carbon-chlorine bond differentiates PCTFE's properties; primarily, the larger chlorine atom in PCTFE disrupts the polymer helix formation that is present in PTFE, effectively lowering the crystallinity and melting point of the chlorofluoropolymer. As crystallinity is lowered, the fluorine atoms sheath the carbon atoms less efficiently, causing PCTFE to be more susceptible to swelling in halocarbon, ether, ester, and aromatic solvents. Though PCTFE’s lower crystallinity makes it more susceptible to chemical attack, it also increases its rigidity, strength, and processability relative to PTFE.
 Gardiner, J. Fluoropolymers : Origin , Production, and Industrial and Commercial Applications. Aust. J. Chem, 2015, 68, 13–22.
Gardiner, J. Fluoropolymers : Origin , Production, and Industrial and Commercial Applications. Aust. J. Chem, 2015, 68, 13–22.
| (2) |
Once in its polymerized state, PCTFE is optically transparent. It is an attractive material for electrical insulation because it maintains its behavior through thermal cycling and high humidity due to its high compressive strength and low gas permeability.
4.2Practical Use as a Shroud Material
When selecting an electrode shroud material, researchers must consider the chemical compatibility, operating temperature range, and mechanical properties of the shroud and their system of study.
4.2.1Chemical Compatibility
A researcher may choose to work with an electrode that has a PCTFE shroud if they are working with reactive and corrosive materials, aqueous or non-aqueous systems. Scientists may also choose to work with PCTFE-shrouded electrodes if their experiments require a highly compressive material.
4.2.2Thermal Stability
Compared to PTFE, PCTFE melts at a relatively lower temperature (~210°C) and has similar low linear thermal expansion coefficients at moderate temperatures (see Fluoroplastics
 Ebnesajjad, S. Fluoroplastics, Volume 1, Non-Melt Processible Fluoropolymers – The Definitive User’s Guide and Data Book, 2nd ed. Elsevier, 2015.
for more information). Like PTFE, an electrode with a PCTFE shroud cannot be thermally cycled outside the small temperature range of 10°C to 25°C (see section 3.2.2). As part of the quality assurance protocol, Pine Research tests all fixed disk/ring electrodes at ambient (room) temperature and has approved PCTFE-shrouded electrode use from 10°C to 25°C. Exposing the electrode to temperatures outside this range may create a leak between the electrode material and the shroud and is not recommended by Pine Research.
Ebnesajjad, S. Fluoroplastics, Volume 1, Non-Melt Processible Fluoropolymers – The Definitive User’s Guide and Data Book, 2nd ed. Elsevier, 2015.
for more information). Like PTFE, an electrode with a PCTFE shroud cannot be thermally cycled outside the small temperature range of 10°C to 25°C (see section 3.2.2). As part of the quality assurance protocol, Pine Research tests all fixed disk/ring electrodes at ambient (room) temperature and has approved PCTFE-shrouded electrode use from 10°C to 25°C. Exposing the electrode to temperatures outside this range may create a leak between the electrode material and the shroud and is not recommended by Pine Research.
4.2.3Mechanical Properties
Hardness and modulus are two common mechanical properties that researchers should consider before selecting a shroud material. PCTFE is a hard material, with a Shore D (durometer) hardness between 85- 95; its exact value is influenced by temperature and resin composition, as well as other factors.
 Ebnesajjad, S. Fluoroplastics, Volume 1, Non-Melt Processible Fluoropolymers – The Definitive User’s Guide and Data Book, 2nd ed. Elsevier, 2015.
PCTFE shroud material is not easily damaged, scraped, or dented. However, because the PCTFE shroud material may not wear down as quickly as the working electrode material, alternative polishing methods, such as a machine polisher, may be necessary to maintain coplanarity of the disk/ring and shroud in the long run. PCTFE has a moderate tensile modulus of 1.4 GPa and could be used in some mechanical stress applications.
Ebnesajjad, S. Fluoroplastics, Volume 1, Non-Melt Processible Fluoropolymers – The Definitive User’s Guide and Data Book, 2nd ed. Elsevier, 2015.
PCTFE shroud material is not easily damaged, scraped, or dented. However, because the PCTFE shroud material may not wear down as quickly as the working electrode material, alternative polishing methods, such as a machine polisher, may be necessary to maintain coplanarity of the disk/ring and shroud in the long run. PCTFE has a moderate tensile modulus of 1.4 GPa and could be used in some mechanical stress applications.
 Ebnesajjad, S. Fluoroplastics, Volume 1, Non-Melt Processible Fluoropolymers – The Definitive User’s Guide and Data Book, 2nd ed. Elsevier, 2015.
Ebnesajjad, S. Fluoroplastics, Volume 1, Non-Melt Processible Fluoropolymers – The Definitive User’s Guide and Data Book, 2nd ed. Elsevier, 2015.
5Polyether Ether Ketone (PEEK)
5.1Physical Properties
PEEK is a semi-crystalline organic thermoplastic polymer that can be produced through multiple methods (electrophilic substitution, nucleophilic substitution, ring-opening polymerization, etc.) from varying monomer sources.
 Shukla, D.; Negi, Y. S.; Uppadhyaya, J. S.; Kumar, V. Synthesis and Modification of Poly(ether ether ketone) and their Properties: A Review. Polym. Rev., 2012, 52(2), 189–228.
Once in its polymerized state, PEEK is a gray/tan solid. PEEK’s aromatic rings and ketones combine to form a very rigid and strong polymer. It is an attractive material for electrical insulation because it maintains its behavior through thermal cycling and high water pressure due to its high compressive strength and low gas permeability.
Shukla, D.; Negi, Y. S.; Uppadhyaya, J. S.; Kumar, V. Synthesis and Modification of Poly(ether ether ketone) and their Properties: A Review. Polym. Rev., 2012, 52(2), 189–228.
Once in its polymerized state, PEEK is a gray/tan solid. PEEK’s aromatic rings and ketones combine to form a very rigid and strong polymer. It is an attractive material for electrical insulation because it maintains its behavior through thermal cycling and high water pressure due to its high compressive strength and low gas permeability.
5.2Practical Use as a Shroud Material
When selecting an electrode shroud material, researchers must consider the chemical compatibility, operating temperature range, and mechanical properties of the shroud and their system of study.
5.2.1Chemical Compatibility
PEEK is highly resistant to both aqueous and organic environments and can withstand other aggressive environments. At room temperature, PEEK dissolves in concentrated acid solutions. At higher temperature, a PEEK shroud can be attacked by some strong Lewis and Brønsted acids and halogenated compounds (though the temperatures needed for this attack are higher than the recommended electrode temperature described in section 5.2.2).
5.2.2Thermal Stability
PEEK melts at high temperature (343°C). In comparison to PTFE and PCTFE, it has superior linear thermal expansion coefficients at temperatures between 10°C and 80°C. As part of the quality assurance protocol, Pine Research tests all fixed disk/ring electrodes at ambient (room) temperature and has approved PEEK-shrouded electrode use from 10°C to 80°C. Exposing the electrode to temperatures outside this range may create a leak between the electrode material and the shroud.
5.2.3Mechanical Properties
Hardness and modulus are two common mechanical properties that researchers should consider before selecting a shroud material. PEEK is a hard material, with a Shore D (durometer) hardness value of ~85; its exact value is influenced by temperature and resin composition, as well as other factors. PEEK shroud material is not easily damaged, scraped, or dented. However, because the PEEK shroud material may not wear down as quickly as the working electrode material, alternative polishing methods, such as a machine polisher, may be necessary to maintain coplanarity of the disk/ring and shroud every so often. PEEK has a high tensile modulus of 4.3 GPa and could be used in most mechanical stress applications.
 Bearing Works, Inc. PEEK (Polyetheretherketone). https://www.bearingworks.com/uploaded-assets/pdfs/retainers/peek-datasheet.pdf (accessed Aug 31, 2016).
Bearing Works, Inc. PEEK (Polyetheretherketone). https://www.bearingworks.com/uploaded-assets/pdfs/retainers/peek-datasheet.pdf (accessed Aug 31, 2016).
6Overall Shroud Material Comparison
When choosing between shroud materials, researchers should first identify necessary and then desirable material characteristics (see Table 2). While the information displayed in Table 2 is not quantitative, it is meant to serve as a guide for basic selection criteria.
For example, a researcher who wishes to perform an experiment in a corrosive environment like concentrated sulfuric acid is limited to using an electrode with PTFE or PCTFE shroud material. If it is important to minimize costs, the researcher should choose a PTFE shroud. If the researcher’s application requires non-standard electrode shroud shapes (such as specific molds) and the ability to withstand large mechanical stress, the researcher should choose a PCTFE shroud.
Additional information can be found in the resources cited in the References at the end of this work (see Section 7).
| Property | PTFE | PCTFE | PEEK |
| Chemical Incompatibility | Alkali metals | Alkali metals, chlorinated solvents, tetrahydrofuran | Concentrated sulfuric and nitric acids |
| Color | White | Optically transparent | Gray/tan |
| Cost | Low | Moderate | High |
| Hardness | Low | High | High |
| Modulus | Low | Moderate | High |
| Creep Resistance | Low | High | High |
Table 2. Property Comparison Between Shroud Materials
7References
- DuPont, Teflon PTFE fluoropolymer resin DuPont: Wilmington, DE, 1996.
- Gardiner, J. Fluoropolymers : Origin , Production, and Industrial and Commercial Applications. Aust. J. Chem, 2015, 68, 13–22.
- Ebnesajjad, S. Fluoroplastics, Volume 1, Non-Melt Processible Fluoropolymers – The Definitive User’s Guide and Data Book, 2nd ed. Elsevier, 2015.
- Shukla, D.; Negi, Y. S.; Uppadhyaya, J. S.; Kumar, V. Synthesis and Modification of Poly(ether ether ketone) and their Properties: A Review. Polym. Rev., 2012, 52(2), 189–228.
- Bearing Works, Inc. PEEK (Polyetheretherketone). https://www.bearingworks.com/uploaded-assets/pdfs/retainers/peek-datasheet.pdf (accessed Aug 31, 2016).



