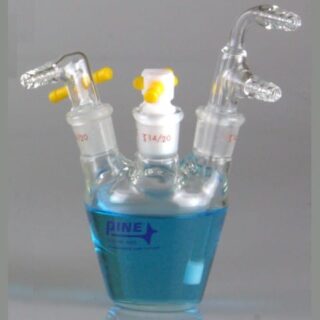
This new platinum counter electrode kit features a 6.6 mm OD PTFE body and terminates with a coil of 0.5 mm OD platinum (Pt) wire featuring an approximate surface area of 4.7 cm². The Pt coil electrode is seated inside a fritted glass tube by means of a 7 mm OD 14/20 PTFE adapter. This fritted isolation tube has a 12 mm OD to 14/20 adapter such that the electrode and fritted tube can pass through a 14/20 port and seal within the ground glass joint. Both adapters enable adjusting the immersion depth of the electrode and isolation tube. The fritted glass tube isolates the counter electrode from the main test solution. This counter electrode is ideally designed for use with our Standard volume electrochemical cells, but is not limited to use in these cells. Customers who have no desire for the fritted isolation tube may consider using this product by itself, as fritted glass tube is not required for use and can be removed if desired. The platinum counter electrode can fit within a 14/20 port directly using its own adapter (included).
MSR evo and RRDE Electrode
MSR evo and WaveNow Wireless
WaveVortex 10 and WaveNow Wireless
MSR evo and WaveNow XV
WaveVortex 10 and WaveNow XV
WaveVortex 10 and RDE Electrode
WaveVortex 10 and WaveDriver 40
MSR evo and RDE Electrode
MSR evo and WaveDriver 40
MSR evo and WaveDriver 100
WaveVortex 10 and WaveDriver 100
MSR evo and WaveDriver 200
WaveVortex 10 and WaveDriver 200
Ungrouped
Electrochemical Cell
Counter Electrodes
Counter Electrodes
- Wang, H.; He, J.; Zhou, M.; Xia, Y. Continuous-Flow and Scalable Synthesis of Pd@PtnL Core–Shell Nanocrystals with Enhanced Activity toward Oxygen Reduction. The Journal of Physical Chemistry C 2024, 128, 21310-21316.
- Ahmed, S.I.U.; Sankarasubramanian, S. Low pH Titanium Electrochemistry in the Presence of Sulfuric Acid and its Implications for Redox Flow Battery Applications. J. Electrochem. Soc. 2024, 171, 060538.
- Vishnosky, N. Synthesis, characterization, and evaluation of metal complexes with cancer selective anti-proliferative effects and hydrogen evolution catalytic properties. Ph.D. Dissertation, University of Louisville, 2019.
- Chen, Z.; Kronawitter, C.X.; Yang, X.; Yeh, Y.; Yao, N.; Koel, B.E. The promoting effect of tetravalent cerium on the oxygen evolution activity of copper oxide catalysts. Phys. Chem. Chem. Phys. 2017, 19, 31545-31552.